Andrea Bellelli
Dipartimento di Scienze Biochimiche A. Rossi Fanelli
Sapienza Universita' di Roma
1. THIOREDOXIN (Trx) is a small, redox-active protein that uses a couple of Cys residues as a two-electron redox center.
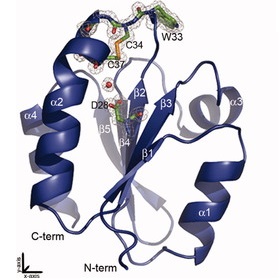
Structure of Thioredoxin from the human parasite Schistosoma mansoni (Boumis et al. 2011, Protein (Structure Report) 20, 1069-1076)
Thioredoxin plays several physiological roles, some of them essential for life, e.g.:
a) it is the obligate electron donor for the enzyme RIBONUCLEOTIDE REDUCTASE (RNR), which converts ribonucleotides to deoxyribonucleotides. No known organism has a direct biosynthetic pathway for deoxyribonucleotides, thus a constant supply of reduced Trx is required for DNA biosynthesis.
b) It is the electron donor of PEROXIREDOXINS (thioredoxin-peroxidases, Prx), which play a major role in the protection against oxidative damage. For example Prx is the second most abundant protein in the erythrocyte's cytoplasm after hemoglobin.
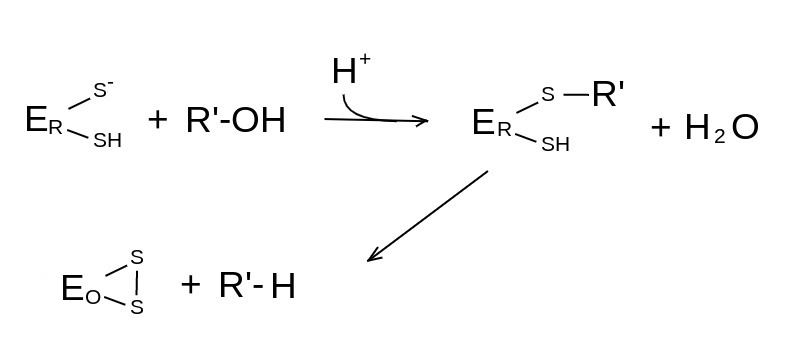
The general catalytic mechanism of Cys oxidoreductases is as follows:
2. Oxidized thioredoxin is restored to its reduced state by THIOREDOXIN REDUCTASE (TR, TrxR) a class of homodimeric enzymes that contain FAD, a redox-active Cys-Cys couple and usually a Sec-Cys couple (in some cases replaced by a second Cys-Cys couple) per subunit.
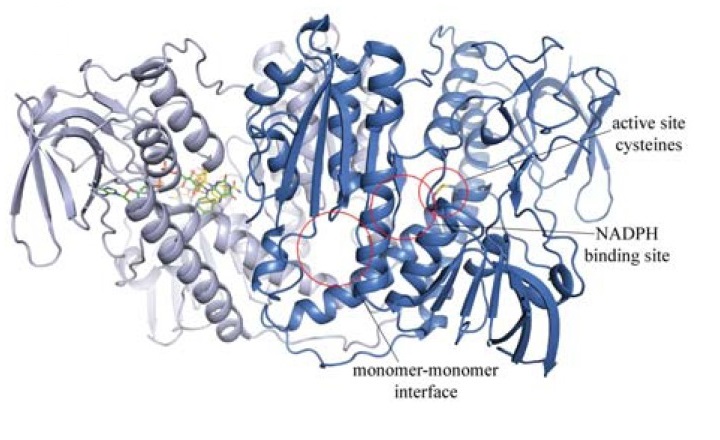
Structure of TGR from S. mansoni. From Saccoccia et al. Curr Protein Pept Sci. 2014;15: 621-46.
In some eukaryotic cells a special form od TrxR is present, capable of reducing both Trx and Glutathione; THIOREDOXIN GLUTATHIONE REDUCTASE (TGR). Remarkably, some parasites (e.g. Schistosomes) possess only TGR, i.e. their ability to reduce both glutathione and Trx depends on a single enzyme.
3. The catalytic mechanism of thioredoxin reductases (and similar oxidoreductases)

Two important observations:
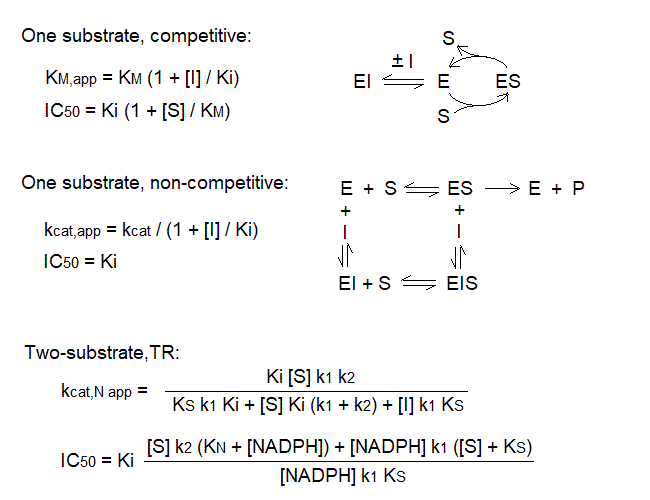
i) KM and kcat, as measured in a typical experiments, i.e. at a constant concentration of either substrate and varying the other substrate, are not constants, because they depend on the concentration of the invariant substrate. One might try to make this substrate high enough to be saturating, but this is not always easy to obtain.
ii) The specificity constant, kcat / KM is a true thermodynamic parameter, independent of the concentration of the invariant substrate.
A typical example of Michaelis parameters for the recombinant thioredoxin reductase from Plasmodium falciparum is as follows: KM for DTNB = 0.24 mM and kcat = 14.7/s, measured at 0,3 mM NADPH in 0.1 M phosphate buffer pH = 7.0, 2 mM EDTA and t=20oC [Boumis et al. 2012 BBRC 425, 806-811]. TR from Plasmodia is interesting and specially suitable for recombinant biosynthesis because it replaces the C-terminal Sec-Cys redox couple with a Cys-Cys couple (Cys535 and Cys 540 in P. falciparum TR).
The electron flow in TR and related enzymes is as follows:
In TRs domain swapping occurs and the C-terminal Sec-Cys couple of each monomer is reduced by the proximal Cys couple of the contralateral monomer in the dimer.
4. There is great interest in TR inhibitors, as they could block or slow down all redox reactions in which Trx is involved. Some Trx inhibitors are in use as drugs for such diverse diseases as cancer, chronic inflammatory and autoimmune conditions, and parasitic infections.
5. Rapidly binding, reversible inhibitors
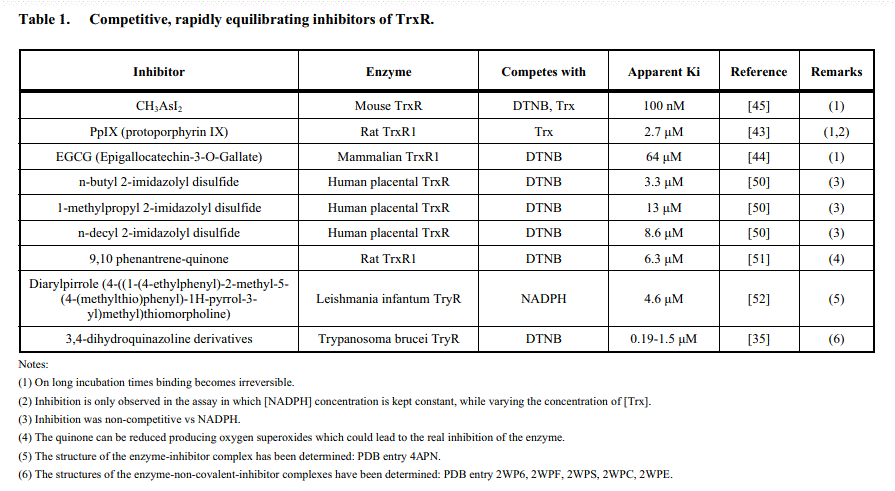
Due to the high reactivity of Cys and Sec, several inhibitors target these residues and are irreversibly or pseudo-irreversibly bound (see below); truly reversible inhibitors of TR are a minority. An interesting exampe is provided by dimethylarsonous diiodide ((CH3)2AsI2), which binds rapidly and reversibly and compete with the oxidizing substrate (Trx or DTNB); however, if allowed a sufficient incubation time binding becomes irreversible. This inhibitor yields typical competitive Lineweaver and Burk plots in experiments run at constant [NADPH] and variable [S], and atypical uncompetitive plots in experiments run at constant [S] and variable [NADPH]:
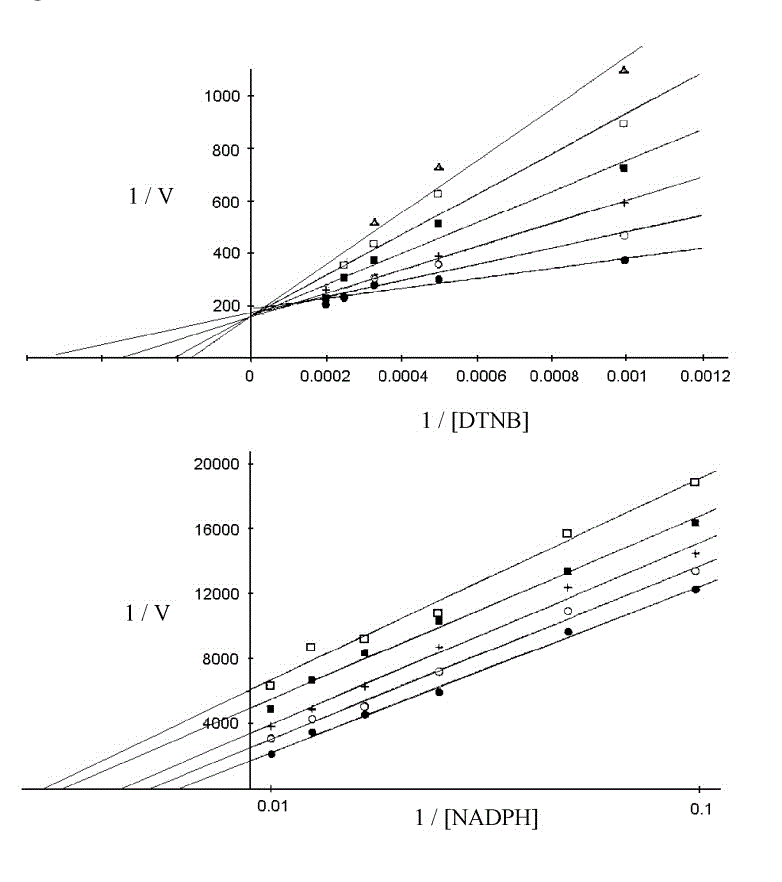
Inhibition of TR from rat liver by dimethylarsonous diiodide, Lin et al. 1999, Chem. Res. Toxicol. 12, 924-930.
The interpretation of these data is not always straightforward (indeed Lin et al. report Ki = 100 nM at variable [DTNB] and 250 nm at variable [NADPH]!). This occurs because the relatioships between Ki and the pertinent catalytic parameters or the IC50, the free inhibitor concentration required to halve the velocity observed in the absence of the inhibitor, differ from the textbook ones (referred to one substrate enzymes), and should be derived for the specific catalytic scheme used:
6. Irreversible inhibitors
Irreversible inhibitors usually target Sec or the catalytic Cys residues. A case in point is provided by Auranofin, a gold-containing metalloorganic complex capable of transferring the metal to the Sec-Cys couple of TR, and via this initial complex to the Cys-Cys couple also. Specificity is kinetically determined, and is provided by the ability of Se to undress gold from Auranofin:
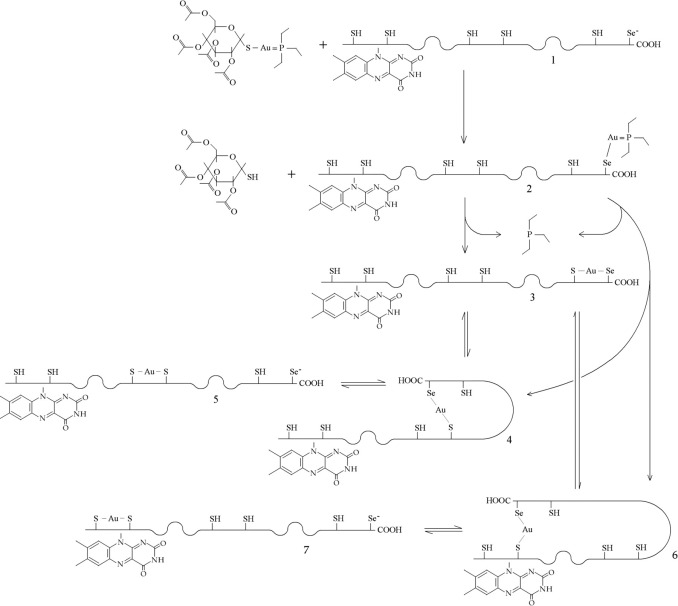
Mechanism of inhibition of S. mansoni TGR by auranofin. From Angelucci et al. 2009, J Biol Chem. 284: 28977-28985
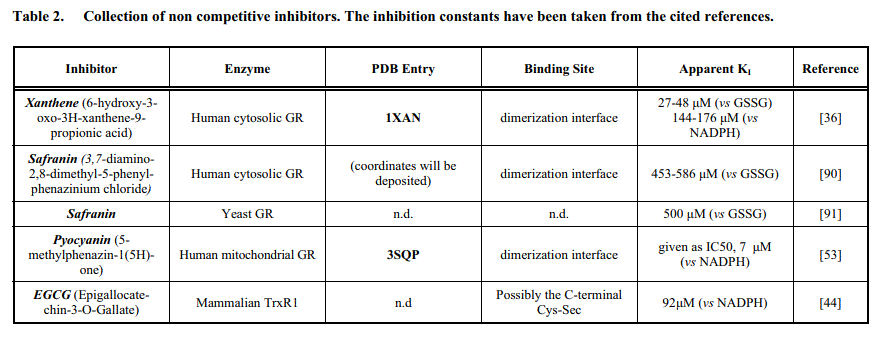
Irreversible inhibitors do not change the KM nor the kcat for either substrate but reduce the concantretaion of the active enzyme [Etot]; as a consequence they lower the Vmax for both substrates (Vmax = [Etot] kcat). Thus they behave as typical non-competitive inhibitors.
7. Slowly binding, possibly reversible inhibitors
Slowly binding inhibitors require an incubation with the enzyme; they typically behave as irreversible inhibitors, because equilibrium with the enzyme derivatives and the substrates does not occur during the time of the essay. Because of this reason distinguishing a slowly binding inhbitor from an irreversible one is difficult and usually requires dedicated experiments. An intermediate case occurs when the rate of binding and dissociation of the inhibitor, while not high enough to reach equilibrium during the essay, is however in the same time range; in this case one obtains curved initial velocity plots. I never observed this case with TRs.
8. Pseudo-reversibility
Pseudo reversibility occurs frequently with TRs: the inhibitor is not strictly irreversible as it dissociates from the enzyme; however it dissociates in a different form from the original chemical species. Often pseudo-reversibility involves the oxidizing substrate as well (Trx, DTNB). The typical reaction scheme observed with alkylating agents is as follows:
TR-Se-Alkyl + Trx-SH → TR-SeH + Trx-S-Alkyl
In this example, which is actually observed for several inhibitors of TR, activity is recovered and the functional state TR-SeH is restored, but the inhibitor is not dissociated in its original form, rather it is transferred to the reduced substrate.
Colleagues and collaborators
Adriana Erica Miele (now at Universite' de Lyon, FR)
Giovanna Boumis (Sapienza)
Francesco Angelucci (now at Universita' dell'Aquila)
Fulvio Saccoccia (now at IBP - CNR, Monterotondo, RM)
David Williams (Rush University, Chicago, IL, USA)